A375-iRFP-Neo
- Frozen / Standard (CL088-STAN) $ 1,500
Species: Human
Cell type: Melanoma
Transgenes: Near-infrared fluorescent protein (iRFP; excitation/emission: 690/713nm) with neomycin resistance (neo) for selection with G418
Media: DMEM, 10% FBS, 1% Pen/Strep, 0.6 mg/mL G418
Description: A375-iRFP-Neo is a polyclonal population of the human malignant melanoma cell line A375 (ATCC® CRL-1619™) transduced with LV-iRFP-P2A-Neo (LV033) encoding the near-infrared fluorescent protein (iRFP) cDNA under the spleen focus-forming virus (SFFV) promoter and the puromycin resistance gene (Puro) under the mouse phosphoglycerate kinase (PGK) promoter.
The lentiviral vector used is a self-inactivating (SIN) vector in which the viral enhancer and promoter has been deleted. Transcription inactivation of the LTR in the SIN provirus increases biosafety by preventing mobilization by replication competent viruses and enables regulated expression of the genes from the internal promoters without cis-acting effects of the LTR (Miyoshi et al., J Virol. 1998).
Mycoplasma Testing: The A375-iRFP-Neo cell line has been tested for mycoplasma contamination and is certified mycoplasma free.
Cell Line Authentication: The parental A375 cell line was authenticated are certified free of interspecies cross-contamination by short tandem repeat (STR) profiling with 9 STR loci including CSF1PO, D13S317, D16S539, D5S818, D7S820, TH01, TPOX, vWA and sex chromosome marker Amelogenin.
Recommended uses:
In vitro: This is a high iRFP expressing cell line suitable for use as a positive control cell line in fluorescence assays to verify iRFP expression in your lentiviral transduced cells.
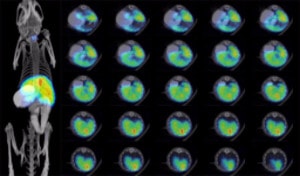
In vivo: A375 cells form tumors post implantation into immunosuppressed mice. The in vivo growth of these tumors may be monitored using noninvasive optical imaging.
Morphology: Low- and high-density cell morphology (200X)

Flow Cytometry for iRFP: A375-iRFP-Neo (red) or control (A375-Fluc-Puro; grey) cells were fixed with paraformaldehyde and analyzed by flow cytometry (20,000 events).

Note: If high resolution nonivasive imaging in living animals is needed, NIS reporter gene is preferred for detection of metastases.
References on NIS imaging:
1. Fruthwirth et al. A whole body dual modality radionuclide optical strategy for preclinical imaging of metastasis and heterogeneous treatment responses in different. microenvironments. J. Nucl. Med 2014. 55(4): 686-94.
2. Penheiter et al. The sodium iodide symporter (NIS) as an imaging reporter for gene, viral and cell-based therapies. Curr Gene Ther. 2012, 12(1):33-47.